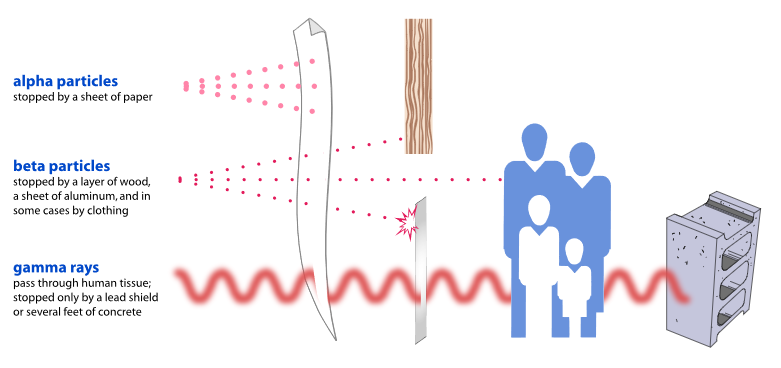
The Radiological Health Program evaluates and manages the actual and potential public health impacts on Vermonters from activities at the Vermont Yankee Nuclear Power Station, located in the town of Vernon in Windham County.
Environmental Surveillance and Monitoring Reports
The Health Department continuously measures the radiation dose around Vermont Yankee, both at the site boundary as well as locations throughout Windham County. On a regular basis, samples are collected around the power station to test for radioactive contamination. Samples are taken from:
-
Air
-
Groundwater
-
Surface water
-
Soil
-
Vegetation
-
Connecticut River sediment
-
Fish from the Connecticut River
Local farms provide milk samples every month for testing to verify there are no radiological contaminants in the milk. Analysis of samples is done at the Health Department Laboratory.
Once every month, the Health Department Laboratory tests private drinking water supplies of selected locations near the Vermont Yankee site boundary. To date, none of these wells have shown contamination with tritium or other radionuclides that would be associated with a nuclear reactor.